
Icosavax
The issue
Our immune system reacts strongly to viruses with a repetitive, symmetric arrangement of proteins. Thus, in designing vaccines, the same idea can be employed, using an antigen with a repetitive, symmetric array to elicit a strong immune response.
Self-assembling proteins, or virus-like particles (VLPs), enable high-density, multivalent display of antigens in a manner that closely resembles viruses, with an important difference. VLPs contain no genetic material, so they are non-infectious and can provide a safer alternative to live-attenuated or inactivated vaccines. VLP vaccines have the potential to be best-in-class, next-generation vaccines as they may deliver safe, effective, and durable protection with one or two doses.
Naturally occurring VLPs have delivered successful vaccines against human papillomavirus (HPV) and Hepatitis B. However, VLPs have been difficult to use for the display of complex heterologous antigens, like in the case of RSV.
The spark
During his postdoctoral research, Neil King, researcher at the Institute for Protein Design (IPD) and an assistant professor of biochemistry at the UW School of Medicine, pioneered the development of general computational methods for designing self-assembling proteins with atomic-level accuracy. His group is extending these methods to design functional protein nanomaterials for applications in the targeted delivery of biologics and the design of next-generation vaccines.

Icosavax cofounders David Baker (left), director of the UW Institute for Protein Design, and Neil King.
The answer
Neil King, along with David Baker, director of the UW Institute for Protein Design, came up with a general computational method to solve the problem of constructing and manufacturing virus-like particles (VLPs) displaying complex antigens. This led to the creation of Icosavax. Icosavax went public in July 2021 and has raised more than $150 million from private investors since 2017.
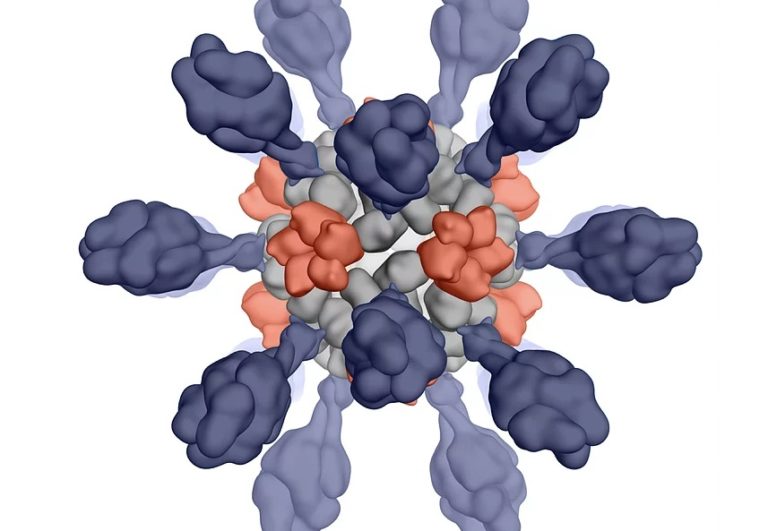
How it works
The breakthrough technology deploys computational methods to predict how proteins behave and fold into specific shapes and generates computationally designed proteins that separate the folding of individual protein subunits from the final macromolecular structure. The individual proteins are expressed and purified using traditional recombinant technologies, and when mixed, they self-assemble into VLPs. The technology enables Icosavax to manufacture VLPs displaying complex antigens at scale to target significant unmet medical needs.
The learnings
Although the original motivation was in biologics delivery, King and Baker pivoted when they recognized how their technology platform would benefit vaccine technology.
“It was this really interesting scenario where the technology platform itself was preconceived and we had a plan for that, but then the application that we actually went after ended up being a hard right turn. I had to make the call between staying on the path and going after biologics delivery, or pivoting into this area of research that I never considered.”
- Neil King, Icosavax co-founder and chair of the Scientific Advisory Board

The UW & CoMotion Boost
In this video, King speaks with CoMotion Director and UW Vice Provost for Innovation, François Baneyx, about the commercialization journey he and his team have been on – the pivots, the strategies, the challenges, and successes.
CoMotion resources supporting Icosavax
- Assistance with patenting and licensing
- CoMotion DubPitch
CoMotion resources supporting Icosavax
- Assistance with patenting and licensing
- CoMotion DubPitch
“CoMotion was instrumental to the entire thing. We filed patent applications through CoMotion, and of course I had never done that before. It is a totally new world and a very different world than protein design or protein structure. I feel like I have gotten an education in intellectual property from my colleagues at CoMotion.”
- Neil King, Icosavax co-founder and chair of the Scientific Advisory Board

The impact
Icosavax is in the process of conducting vaccine trials for respiratory syncytial virus (RSV), human metapneumovirus (hMPV), and SARS-CoV-2. These vaccines have the potential to induce high neutralizing antibody titers and could protect the most vulnerable populations, including older adults, against severe respiratory diseases. RSV is a major cause of viral pneumonia, and there are currently no specific antiviral therapies or vaccines for RSV. Additionally, the IPD is applying the designed nanoparticle platform to global health targets, such as malaria.